Choosing the correct floor cleaning chemical can make a major impact on the effectiveness of your floor maintenance program. Floors that are plagued with streakiness, for example, are likely being cleaned with the wrong detergent. The pH of the cleaner is an important factor in determining which chemical is best for your application.
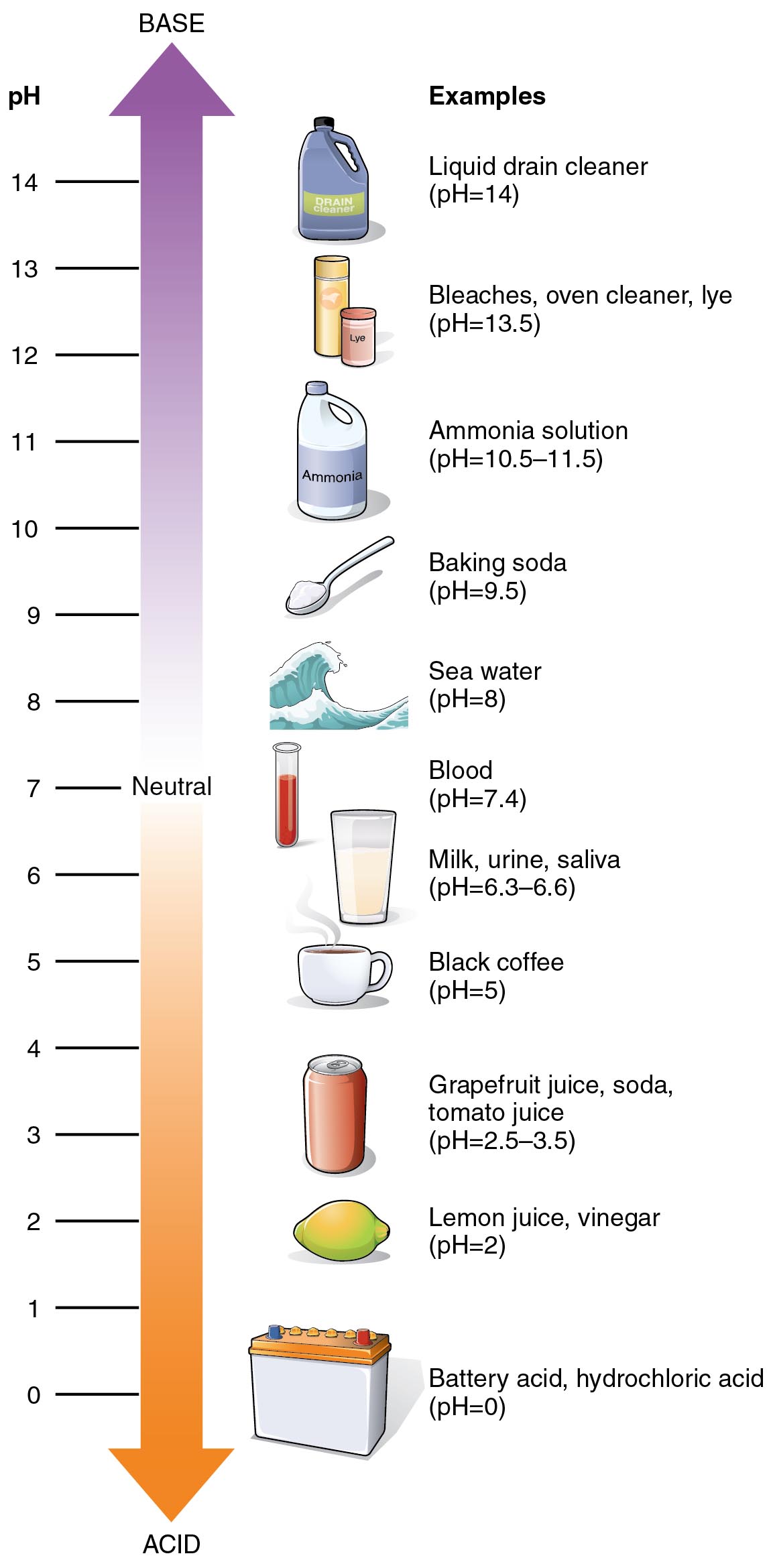
The pH scale ranges from zero to 14, with a pH of seven being neutral. A cleaner ranging from zero through six on the pH scale is an acidic detergent and a cleaning chemical ranging from eight to 14 on the pH scale is an alkaline detergent. The pH level of a cleaning chemical should not be confused with the chemical’s strength. This number is a gage of the concentration of hydrogen ions or H+, not an indictor of performance power.
Neutral Cleaner
A neutral pH cleaner is usually the ideal choice for general purpose daily cleaning of surfaces that are not heavily stained or soiled. Water also carries a neutral pH, so essentially any floor material or finish that can be cleaned with water can also be cleaned with a neutral pH floor chemical without damage. This type of floor cleaner is also one of the most environmentally friendly options. Clean and Green detergent is one example of an environmentally friendly neutral pH floor cleaning chemical that was formulated with the US EPA and can be used on almost any floor surface, for a wide range of applications, in a scrubber or a bucket, without leaving residue behind. Gentle enough for waxed surfaces, a neutral cleaner is safe and effective for a wide variety of floor finishes.
Acidic Cleaner
An acidic floor detergent is the choice chemical to remove rust stains, mineral deposits, and heavy organic matter. Vinegar and lemon juice are two acidic substances that are often used for cleaning purposes. Removing hard water spots, tarnish, and calcium build-up should be done with an acidic floor chemical. Marble, travertine, and limestone are a few surfaces that should never be cleaned with an acidic cleaner.
An acidic floor chemical can be used as a neutralizer, which means that it can be used to bring alkaline spills or residues back to a pH of 7 or neutral. One example of using an acidic detergent as a neutralizer is when using it to remove salt. Winter months can wreak havoc on floors because the salt that is used to melt ice is a highly alkaline material which can be destructive to floor finishes. The alkaline pH of salt makes using a neutral pH general purpose cleaner on that substance less effective than an acidic detergent. An acidic floor chemical will neutralize the floor, removing the alkaline salt much more effectively than the pH neutral cleaner.
Alkaline Cleaner
If floor stripping is desired, a highly alkaline floor chemical is likely the best chemical for the job. An alkaline cleaner that is closer to neutral on the pH scale is often a good choice for general purpose cleaning or degreasing. Alkaline cleaners can be used to remove grease, animal fats, and oil stains. Bleach and ammonia are two examples of alkaline substances that are often used for cleaning.
Like acidic cleaners, alkaline floor detergents can be used as neutralizers. An alkaline chemical can effectively remove acidic spills or residues, bringing the floor back to a neural pH of seven. On concrete, for example, an alkaline cleaner is the best option for use following an acid staining or acid cleaning. Restoring tile and grout should also be done with an alkaline cleaning chemical.
Understanding the pH of the floor chemical that you are using is key to determining if it is the most appropriate cleaner for the job. For assistance choosing one of the many quality detergents available online in our detergent store, contact the floor cleaning equipment experts today at 800-793-3790. For more on floor detergent basics, check out “Choosing the Right Detergent” from our cleaning blog.

